Technical Deep Dive: Validating Vacuum Decay CCI Testing for 2 mL & 10 mL Vials
We’re pleased to share a detailed look into our recent validation work on the Bonfiglioli Engineering LT-PRO HA Container Closure Integrity Tester (CCIT), now qualified to support sterile manufacturing operations for both 2 mL and 10 mL vial formats.
This effort represents a critical component of our contamination control strategy and alignment with the expectations of EU GMP Annex 1 and USP <1207> for deterministic container closure integrity testing.
1. Objectives:
The goal was to develop, optimise, and verify vacuum decay leak-test cycles capable of reliably detecting leaks greater than or equal to 1 μm defect size, while avoiding false rejection of known-good product.
As a non-destructive and highly sensitive method, vacuum decay offers:
- High detection capability through differential pressure measurement
- Repeatability and quantitative evaluation
- Suitability for 100% in-process or batch-based testing
- Compliance with regulatory expectations for deterministic CCIT methods
2. System Description:
Equipment: Bonfiglioli LT-PRO HA
Testing method: Vacuum Decay
Formats: 2 mL and 10 mL Type I glass vials
Container closure components:
- SCHOTT adaptiQ® vials
- West FluroTec® Stopper
- No-text flip-off aluminium seals

3. Cycle Development:
Each cycle was developed using:
- Representative “known good” vial assemblies
- A stainless-steel zero-leak reference vessel
- A calibrated leak orifice corresponding to a 1 μm defect — the maximum size considered capable of maintaining sterility
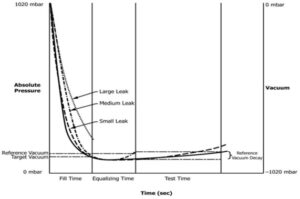
The cycle is broken into defined phases; these are depicted in Diagram 1:
- Pre-Fill Vacuum: Detects gross chamber/system failures and establishes baseline vacuum performance.
- Fill Phase: The chamber is pulled to nearly full vacuum (-970 mbar target), chosen to maximise differential pressure and increase sensitivity. Fill times and equalisation periods were experimentally optimised for both vial types to ensure stable starting conditions.
- Test Phase (Vacuum Decay): Continuous high-resolution monitoring identifies any vacuum loss beyond the allowed decay threshold. The test duration and decay limits were statistically determined to distinguish good samples from 1 μm leaks with ≥99.7% confidence.
- Vent & Sort: Automated venting and identification of passed and failed vials.
Diagram 1 – Vacuum Decay Cycle
3.1. Statistical Evaluation
On completion of cycle development, optimisation was achieved through the statistical evaluation of data from 2 populations of data generated from both known good samples and data utilising a calibrated leak and zero leak test piece to occupy the sample chamber.
For both vial formats, we calculated:
- Mean decay values
- Standard deviation (σ)
- ±3σ boundaries to represent a 99.7% statistical band
A reference decay limit was then selected between the following for each cycle (Diagram 2):
- The known-good +3σ limit
- The calibrated leak −3σ limit
Diagram 2 – Setting Reference Decay
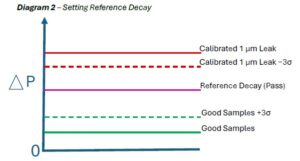
This ensured:
- Zero overlap between populations
- Reliable detection of ≥1 μm leaks
- No false positives against acceptable product variability
4. Key Outcomes
✓ Vacuum decay cycles demonstrated robust separation between intact and defective containers
✓ Sensitivity meets requirements for ≥1 μm defect detection
✓ Good vial variability is fully accounted for — reducing false rejects
✓ Test cycles were fully verified during FAT and further confirmed during SAT, IQ, and OQ
✓ System performance aligns with USP <1207> deterministic CCI principles
✓ Equipment is now ready to support routine CCI testing for our sterile filling operations
Why This Matters
Ensuring container closure integrity is fundamental to patient safety, especially as regulatory expectations continue to increase. Deterministic, validated CCIT methods such as vacuum decay provide:
- Objective, quantifiable assurance of seal integrity
- Improved detection sensitivity vs. probabilistic methods
- Lower operational burden and lower sample destruction
- Critical compliance with modern contamination control frameworks
This validation is a significant step toward strengthening the sterility assurance of products produced in Unit 8.
Hashtags:
#PharmaceuticalEngineering #QualityAssurance #CCIT #VacuumDecay #USP1207 #Annex1 #ValidationScience #SterileManufacturing #Eramol