Clinical Storage & Distribution


Clinical Storage & Distribution Services
Your Trusted Partner for GMP/GDP-Compliant IMP Storage and Distribution Across the UK, EU & Globally
Safe, Efficient, and Compliant IMP Logistics
Eramol provides comprehensive clinical storage and distribution services, ensuring your investigational medicinal products (IMPs) remain secure, stable, and compliant throughout the supply chain. With state-of-the-art temperature-controlled facilities, expert logistics teams, and QP release capabilities, we act as a trusted gateway between the UK and EU for clinical trial materials.
End-to-End Storage and Distribution Expertise
Eramol delivers GMP and GDP-compliant storage for clinical trial materials, offering full accountability and supply chain integrity across all stages of your project:
- Temperature-controlled storage: -80°C, -20°C, 2–8°C, 15–25°C
- Continuous monitoring with uninterrupted power backup
- Controlled substance storage: Schedule 1–5, including medical cannabis (THC/CBD)
- Secure facilities for long-term storage prior to shipment
Our logistics experts coordinate shipments, ensuring your IMPs reach trial sites, patients, and partners on time, safely, and fully compliant.
Global and Local Distribution Capabilities
Eramol provides direct-to-site, direct-to-patient, and comparator supply management through a network of global depots and approved couriers. Our services include:
- UK, EU, and Global IMP hubs with QP release and verification
- IVRS/IWRS integration to support clinical trial management
- Returns, destruction, and comparator management
- Validated cold chain shipping containers
- 24/7 emergency logistics support
Our expert team manages complex regulatory and logistical challenges, enabling seamless clinical supply management.
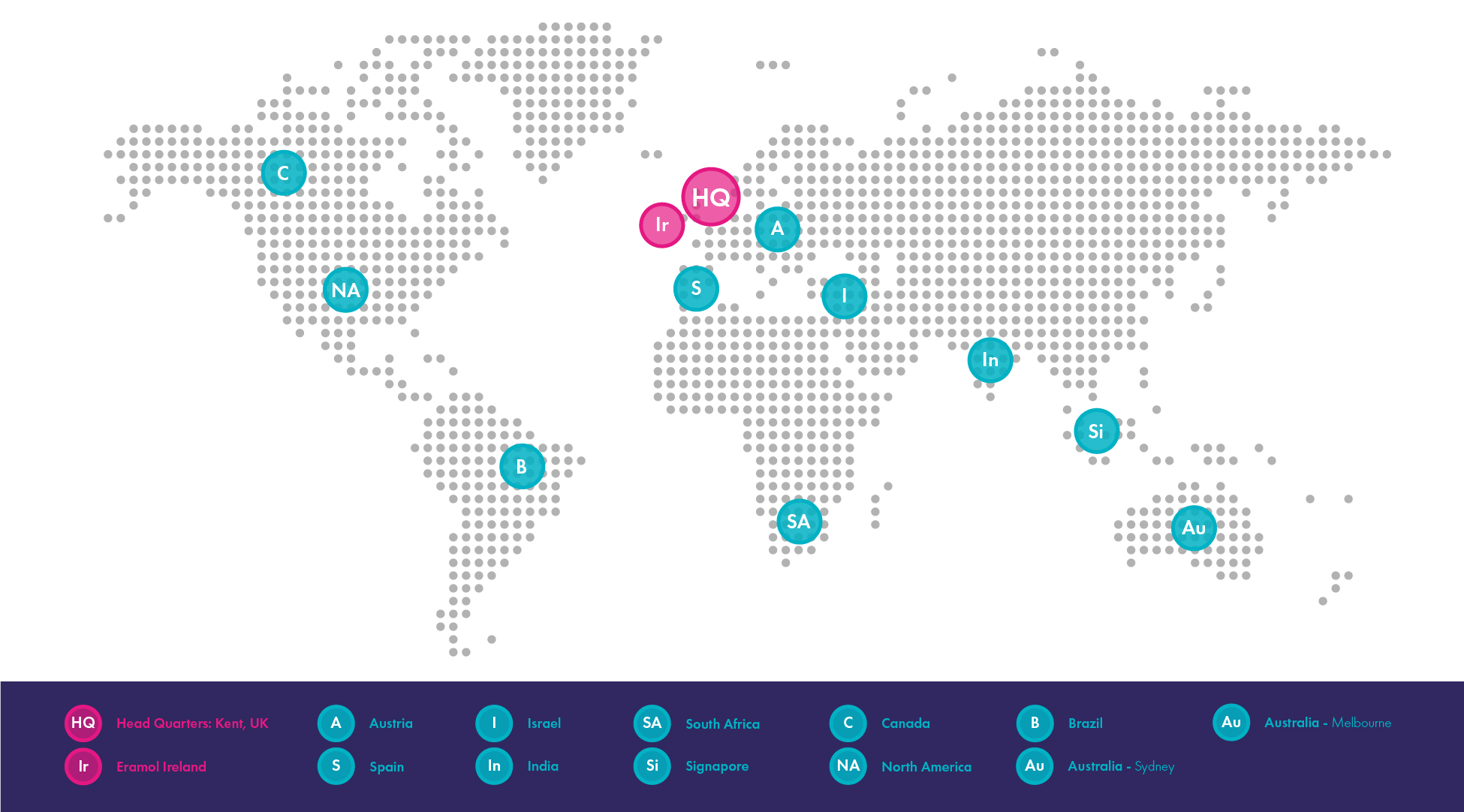
Strategic Locations
Eramol’s network of depots enables efficient distribution worldwide:
Eramol UK (HQ)
- Primary & secondary manufacturing
- Cold chain & ambient storage
- Narcotic storage & distribution
- UK QP Release/Verification
- Global distribution hub
Eramol Ireland (Dublin Facility) – Gateway to Europe
- Physical site of EU import
- EU QP release
- Cold chain & ambient storage
- Direct-to-trial site distribution across the EU
Local EU Depots
- Austria: EU importation, cold chain, controlled substances
- Spain: Local storage & distribution, narcotic handling
- Poland: Cold chain & ambient storage, distribution support
Other Global Depots
- Australia (Melbourne & Sydney)
- North America
- Canada
- Brazil
- Singapore
- India
- Israel
Importer of Record (IOR) & Customs Services
Eramol is an accredited Importer of Record (IOR), managing:
- Compliance with local and international import/export laws
- Customs documentation and duty entry
- Payment of import duties and taxes
- Full traceability and accountability across supply chains
Our IOR expertise ensures smooth, compliant importation into the UK, EU, and global markets.
Gateway Between the UK and EU
Eramol’s dual-site strategy positions us as a key partner bridging UK and EU clinical trials:
- Irish facility: EU importation, EU QP Release, storage, and distribution
- UK facility: QP verification for EU-imported IMPs, local UK distribution
- Solutions for Northern Ireland trial sites via Dublin facility
- Specialist QP regulatory support for cross-border IMP supply chains
Whether you are importing locally, across Europe, or globally, Eramol ensures your IMP supply is compliant, secure, and efficiently delivered.
Why Partner with Eramol
- Trusted UK & EU distribution partner for clinical trials
- Extensive temperature-controlled storage for IMPs
- QP release and verification across all UK and EU sites
- Controlled substance management, including narcotics and medical cannabis
- Global network of depots supporting direct-to-patient and comparator supply
- Expertise in customs clearance, IOR, and regulatory compliance
- 24/7 logistics support for emergency management
Ensure your clinical trial materials are stored, managed, and distributed safely, compliantly, and efficiently.
Reliable. Proven. Safe. Effective.
Reliable. Proven. Safe. Effective.
Speak With Us
Products on online services or over the Internet. Electronic commerce draws on
technologies such as mobile commerce application